- Ron Rosedale – Fiber – Need It or Not?
- The American Gut Project
- Justin Sonnenburg – Fiber & “Starving our Microbial Self”
- Alberto Martin – Carbohydrates and Colon Cancer
- Eric Westman MD – Low Carb, Health & the Microbiome
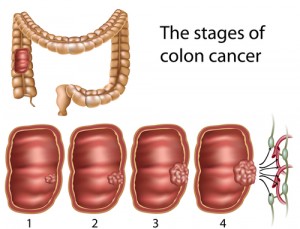
INTRO from Shelley: Many advocates of high fiber diets say that fiber is essential for good digestive health, and that with a high fat, low carb diet fiber tends to be too low. A leading rationale for advocating high-fiber diets is to feed “resistant starch” to the beneficial microbes in the gut, so that they can make the gut cell’s favorite food, which is butyric acid. But could too much butyric acid, be too much of a good thing? Today, we talk with University of Toronto scientist Alberto Martin, about his 2014 study that appeared in the Journal Cell, titled Gut microbial metabolism fuels colorectal cancer in Msh2-deficient hosts.
Martin’s study indicates that mice bred to have a common form of human colon cancer get the cancer more quickly when the mice have lots of butyrate, in their gut. Martin’s team delivered butryate through regular, high fiber mouse chow. They got plenty of cancer. When the researchers fed the mice a low-carb, low-fiber diet, they didn’t get cancer as fast. However, the researchers could tweak the cancer incidence in either direction by manipulating the amount of butyrate in the gut, either by adding butyrate as a supplement, or by killing off butyrate producing microbes with antibiotics. This study was done in mice, but Alberto Martin points out that epidemiological evidence indicates that humans with this kind of genetic mutation get colon cancer more often when they eat a high carb, more high fiber diet. He says about 1 in 80 North Americans gets this kind of colon cancer.
(NOTE – This transcript includes paraphrasing)
SHELLEY – With regards to colon cancer, your new research study indicates that too much available butyrate in the gut can be too much of a good thing.
ALBERTO MARTIN – This is correct. Our data points to this. Our data indicates that with regard to carbohydrate, specifically those who harbor mutations in mis-matched repair genes, a high carbohydrate diet is not a good thing. I want to preempt everything by saying that our studies were done in mice, not in humans, but there is some epidemiological evidence that a high carbohydrate diet in humans is linked to colon cancer.
SHELLEY – Can you say some more about that? After all many proponents of high-fiber diets say that the colon is healthier with carbs.
ALBERTO MARTIN – It all depends. We’re arguing that carbs, with this specific genetic mutation, is not good for colon cancer. We’re not saying that carbs is generally a bad thing. We’re just arguing that if you have this genetic defect, you might want to reconsider eating a high carbohydrate diet. Now it’s possible, and I don’t know enough about this, that a high carbohydrate diet might be good for other things related to intestinal health. But with respect to what we’re studying, it’s not good. When you think about colon cancer being one of the most prevalent cancers in the world. Of those cancers, 20% of all colon cancers harbor these mutations. That’s a large population of the Western population. One in 15 of us will develop colon cancer in our lifetime, and 20% of those will be colon cancer that harbors these types of mutations. So that’s 1 in 80 North Americans who will develop these types of colon cancer.
SHELLEY – One in 80 people will develop this kind of mutation-prone colon cancer, as the mice did. So your mice – they were sitting ducks for cancer because you had bred them specifically to have that double-DNA mutation..
ALBERTO MARTIN – Right.
SHELLEY – You fed them in a way, in a couple of different ways, got normal levels of butyrate into their colons. In one of those ways, they were also eating lots of carbs. Do you think it’s the combination of butyrate and high insulin levels from the carbohydrates that made them more prone to developing cancer? Or is it just the butyrate itself that promoted the cancer?
ALBERTO MARTIN – These studies are still on going. We cannot say that it’s only butyrate. That would be naive to think it’s really only one element from a specific diet – it could be a combination of things. What we’re arguing is that we see a correlation between butyrate levels and colon cancer. Our data suggests that it’s butyrate, but we’re not saying it’s only butyrate.
SHELLEY – Ron Rosedale is a medical doctor and scientist who told me about your study (see interview here) and added that intestinal cells are somewhat unusual in terms of what they eat, because they don’t feed on carbs and sugar, but they love to eat butyrate. They thrive on butyrate.
ALBERTO MARTIN – It’s energy. It’s energy for colon epithelial cells. They require this as energy, and that’s their primary energy source.
SHELLEY – His guess was that it’s like too much of a good thing anywhere else. If you feed most cells more than enough sugar, for instance, their maintenance and repair goes out the window and instead, they go into more rapid proliferation of cells. They divide more often, which is one hallmark of setting things up for cancer.
ALBERTO MARTIN – I would tend to agree with that statement. Obesity is associated with cancers. It’s the Warburg effect. The idea is that cancer requires lots of sugar. Basically they need tons of sugar. They need tons of energy. My feel is that if you eat too much you’re going to predispose yourself to a number of kinds of cancer.
SHELLEY – One of the mechanisms might be simply that if you give your cells too much available food, it overheats them into dividing more rapidly and does not exercise their maintenance and repair mode enough.
ALBERTO MARTIN – Potentially. Right.
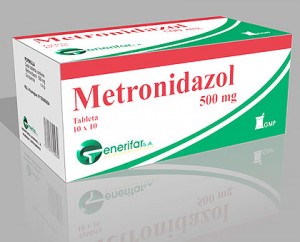
SHELLEY – So you said one way you tested whether it was other components of the mice diet that promoted the cancer, or just that you were feeding them foods that fed the microbes that made lots of butryate, is that you cut back on the butyrate by giving those mice the antibiotic, metronidazole.
ALBERTO MARTIN – To kill off the bacteria that produce butyrate.
SHELLEY – I thought metronidazole kills yeast too.
ALBERTO MARTIN – Yes and it kills a lot of other things. It kills a lot of bacteria and produces a major shift. We’re not suggesting this is a good way forward to prevent the onset of colon cancer. Because in the end bacteria play a very important physiological role for us. We’re just at this point not doing any therapeutic or preventative analysis. We’re just trying to figure out how colon cancer develops in these mice. The better way forward would not be to use antibiotics, because then you develop antibiotic resistance. It’s really better to alter your diet.
SHELLEY – Plus you also mess up the healthy part of the micro biome in all kinds of unknown ways
ALBERTO MARTIN – Right. There are very good microbiota you want to have. Including those that we target with metronidazole. Those we targeted are considered normal microbiota, that have no physiological consequences, except for providing us with extra energy that, for someone with this genetic background, that extra energy is not a good thing. So metronidazole is not a smart bomb. It wipes out a lot of bacteria, including some that are healthy for us.
SHELLEY – So with one arm of your experiment, you killed off the microbes that make the butyrate that the colon cells like to eat, to see whether that would slow down growth of colon cancer. And it did. You also put a group of mice Ton a low carbohydrate diet – which reduced the amount of butyrate produced by the microbes in the colon, and those mice had slowed growth of colon cancer. Then you did a flip of the experiment. With the mice fed a low carb diet, which meant their gut microbes weren’t generating much butyrate, you very intentionally added butyrate as supplement to their low-carb diets, plus you also gave the mice little mouse enemas – I mean suppositories.
ALBERTO MARTIN – That’s a very difficult experiment. We’d have to do enemas for basically three weeks, daily or every other day. What we did there, using the enema experiment was assess whether it produces hyper proliferation of intestinal cells. And then correlate what we saw, when we did the butyrate enemas, regarding the mismatched repair, defective colon epithelial cells, which were now hyper proliferating and then we correlated that to the colon cancer.
SHELLEY – That means you did some tissue analysis of the mice as well. What I was thinking, when I said enemas, actually, was a little different part of what your experiments involved. I was thinking of the suppositories of butyrate that you gave the mice. In one part of your paper, it sounds like you’re saying, that feeding the mice their regular cracked corn, high fiber diet got plenty of food to the colon for feeding the microbes that make butyrate there. But when the mice were fed a low carb diet, and you added supplements of butyrate by mouth, the rest of the body metabolized that dietary butyrate so quickly, it never went down into the colon in a big amount, so you had to use a suppository through the rear end to increase the amount of butyrate that actually arrived in the colon. That’s interesting, too – that dietary butyrate doesn’t reach the colon in any big way. But another question now – to create a diet that’s low carb for the mice, you gave them a ton of protein. It was what – about 70% protein.
ALBERTO MARTIN – If you’re going to remove carbs, you have to add something. What we have not done, or are doing now, is . . . Instead of having a very high protein diet, if you have a high fat diet, at the expense of carbohydrates. That will help us look at – are carbs the only bad thing for this type of colon cancer.
SHELLEY – I’m guessing you used high protein because even though excess protein will turn into ammonia and sugar, at least those nutrients get absorbed so fast, they don’t make it into the gut,. After all, the issue for your study was that you were looking at how dietary fiber and slow-to-digest carbs, end up in the gut to feed the microbes that produce butyrate. The ammonia and sugar from excess protein doesn’t quite make it into the gut.
ALBERTO MARTIN – Sugar doesn’t make it into the gut. It will get absorbed very early on in the small intestines. The only way to deliver sugar, or energy, to the colon is through polysaccharides or carbohydrates. They can’t get absorbed in the small intestine. Once they get to the colon they get broken down by bacteria into sugar and short chain fatty acids. And then they’re absorbed.
SHELLEY – The common way what you just said gets talked about right now is to refer to those nutrients as resistant starches. Starches that don’t get broken down until they get into the colon. But getting back to diet – when you fed the mice 70% protein. Feeding the mice that much protein is not good for them.
ALBERTO MARTIN – They don’t like it too much. They urinate a lot. It’s not a healthy diet. We’re just using extremes here for testing a specific theory.
SHELLEY – Yes, but despite the high-protein diet’s unhealthiness, it didn’t promote colon cancer in your mice as regular mouse chow did, which is high in complex carbs. Now given what you’re seeing, you’re curious about what a high fat diet would do. Would it promote or slow down their risk of colon cancer? And I mean a real, high fat, low carb diet, not just the typical one that’s a moderate amount of fat, paired with a lot of cornstarch and sugar. A truly high-fat, low carb diet is a little tricky in mice because they don’t go into ketosis unless the ratios are just right, and generally it takes even higher fat and lower carb for mice to reach ketosis than it does for humans There’s that really runny high fat mouse that’s commonly used as a way to try, and I’ve heard from other researchers it’s not very successful, because the mice hate it.
ALBERTO MARTIN – When we were raising dietary butyrate by providing mice with a three-butyrate molecule they could eat in their food, they don’t like that because butyrate smells. The high fat diet might be also very difficult, because they don’t like that kind of food.
SHELLEY – When you fed them the three-molecule butyrate, you said it had already been absorbed before any of it reached the colon. Did it get down below the stomach, into the duodenum, or even further, into the small intestine?
ALBERTO MARTIN – In the end, what happened when we fed them butyrate, we were able to increase the levels in the small intestine but not the colon. So it didn’t make it all the way down into the colon. In the mice that we fed this way, we could see increased polyp number in the small intestine, which is an indication that it was inducing colon cancer in the small intestine. But we couldn’t carry out the experiments in the colon because the butyrate wasn’t getting there. That’s why we did the enemas.
SHELLEY – You were looking at only colon cancer or small intestine cancer?
ALBERTO MARTIN – We were looking at both.
SHELLEY – So the extra dietary butyrate that you fed to the mice on a low carb diet, it did induce polyps in the small intestine. One mechanism Ron Rosedale mentioned, as a possibility about why, is that, on a high protein diet, those mice are probably also fairly high in insulin. That’s his guess.
ALBERTO MARTIN – Possibly. We did not test that.
SHELLEY – His guess is, you had two promoters of cancer going. The insulin pathway will push cells to divide and multiply faster, and then there’s the MTOR pathway. Target of Rapamycin. Ron’s thought was, what if the combination of pushing the insulin and MTOP pathways hard with high protein and then adding butyrate, is not so good? But . . . He was curious about what would happen if it was a high fat diet, low carb, adequate protein diet.
ALBERTO MARTIN – It’s a very good idea for an experiment. And we’re doing that, because right now we can’t argue that the high protein diet is protecting the mice from colon cancer. We still need to test whether, by having normal protein and increasing fat and still keeping carbohydrate low, how does that influence the risk of colon cancer.
SHELLEY – Ron’s speculation is that if a creature is adapted to eating a high fat diet, they may metabolize the fats rapidly enough, there’s enough butyrate getting into the colon to provide adequate support to the colon and the small intestine microbes, but not so much that it pushes them into overdrive. If it also keeps the insulin pathway down and the Mtor pathway down, it has potential to be more protective against this colon cancer.
ALBERTO MARTIN – It’s worth testing
SHELLEY – And the challenge is that mice are not high fat eaters, usually. As for the Butyrate that reaches the intestines, it’s okay maybe to have just a little, but not to much of it.
ALBERTO MARTIN – That’s what we’re arguing. Given this genetic background, we’re arguing a certain more “average” amount of butyrate is not good. Interestingly, though, a great deal more butyrate, by using the enemas, it actually has a protective effect. Butyrate actually has these duel activities. At low levels of butyrate, these more physiological concentrations of butyrate, it actually acts as a proliferative agent. But at a very high concentration, butyrate acts at as a Hdac inhibitor. Histone de-acetylase inhibitor. And it inhibits proliferation.
SHELLEY – Very high levels of butyrate inhibit cells from multiplying and dividing extra fast?
ALBERTO MARTIN – That’s a known effect. It acts very differently at very high concentrations. It’s a Histone deacetlyase inhibitor. By inhibiting histone deacetylase it promotes a reduction in proliferation.
SHELLEY – It also promotes more maintenance and repair when it’s super high?
ALBERTO MARTIN – When you say repair, what do you mean by repair?
SHELLEY – I guess what I’ve heard often is that cells kind of are in one of two modes. They’re either conserving their resources and fixing up their shop – you know -sort of hunkering down to make it through a famine, or they’re saying gee there’s lots of energy right now. Must be time to multiply and divide. I’ve always thought of these two modes as flip sides of a coin.
ALBERTO MARTIN – And they could be. I don’t know enough about cellular physiology in the colon with respect to repair and pathways vis a vis repair and pathways for replication and how they interact. I don’t know enough about that.
SHELLEY – In any event, in these mice, one reason you gave gave butyrate suppositories is to see what ultra-high levels of butyrate would do.
ALBERTO MARTIN – Very high levels of butyrate, we saw a reduction in proliferation. So we saw this bi-modal effect.
SHELLEY – So you think it switched the pathway once again.
ALBERTO MARTIN – Very high levels, it acts differently. Butyrate, It no longer acts as an energy source. It starts acting as a modulator of gene transcription by inhibiting histone deacetylase.
SHELLEY – Some researchers have told me the issue isn’t so much that there aren’t enough beneficial microbes in the gut but instead that the beneficial microbes keep the bad bugs in check.
ALBERTO MARTIN – That’s the common thinking. Yes.
SHELLEY – From how you said that, I can’t tell whether you agree with the common thinking or not.
ALBERTO MARTIN – I think it’s the current thinking. I don’t think any of us are in the position to say whether that’s correct or not. We may need a microbiota there to keep the bad guys out. But microbiome researchers are still in the early days. So we’re still learning about how the microbiota influences and is influenced. The term, dysbiotic microbiota, you see an increase in pathogenic microbes. Is that due to a loss of the non-pathogenic microbes, the commensal microbes? We’re still learning a lot about the different interactions, really trying to understand how these communities function together and keep other bacteria in check. That statement, I would say, currently is accurate. But it’s still early days.
SHELLEY – Looking at high fat and the microbiome, in the blogging community, there’s such a rift about whether high fat and lower fiber is good or bad. You as a researcher, I hear you saying, really the jury’s out. We don’t know.
ALBERTO MARTIN – I think we have bits of evidence. The bits of the puzzle are starting to form, but we still haven’t completed the puzzle. There are still surprises.
SHELLEY – You did a very low carb, high protein diet with your mice, which is not the same as a very low carb, high fat diet. In general, a “high-fat” diet for a mouse is adequate protein, plus the rest divided in half between carbs such as sugar and cornstarch, and fat such at lard. That kind of “typical” high fat diet won’t show the same results as a truly high fat-low carb diet. For instance, one researcher said to me, when you’re feeding the carb-loving microbes, you feed both beneficial carb-loving microbes and ones that might aggravate the intestinal lining.
ALBERTO MARTIN – We see major shifts in the bacterial community when you change diets and reduce the carbohydrate. The bacteria that require carbohydrates, they will slowly disappear – not disappear completely but be reduced in numbers.
SHELLEY – How many days does a body need for a shift in diet to make a shift in microbes.
ALBERTO MARTIN – One week is sufficient to see measurable differences. If you cut carbs, you will see measurable difference in one week. If you take a baseline one week later, it might be even more steady. Baseline one week, two weeks, three weeks, each might change a bit more. But you will see measurable differences with a one week shift.
SHELLEY – A week would be enough, but two weeks would be a little better.
ALBERTO MARTIN – We don’t know enough to say for sure. Plus, different diets are likely going to lead to different changes in the microbiota and at different rates, and with different baselines. If you have a high fat diet, the baseline might take even longer or shorter. I don’t know. There might be some scientists, I should add, that have already tested this, and I’m just not aware of the data. I don’t know. And poop samples is one thing, but the bacteria that reside along the mucosal surface of the intestine, in the colon, is different from what comes out in the poop. The bacteria that reside along the mucous interface, there’s more oxygen right next to the tissue lining the gut. So the bacteria residing there tend to be more oxygen resistant than those that exist further into the lumen. The ones in the lumen tend to be more obligate anaerobes, meaning exposure to oxygen can kill them.
SHELLEY – Sounds like you’re especially interested in the microbes that like to snuggle in right next to the intestinal lining where there’s a bit more oxygen.
ALBERTO MARTIN – There’s a bit of oxygen. It’s not going to be as low in oxygen as it is further toward the middle of the lumen. Using poop analysis to measure effects on diet and microbial composition, is just one variable. But If you want to be more complete you want to combine the poop analysis with the mucosally associated bacteria. Typically the ones, as shown in a recent paper by Gary Rube, he shows that the bacteria that basically metabolize carbohydrates are found into the lumen, so further away from the mucosa.
SHELLEY – So the ones in the middle, in the lumen are providing the food, the butyrate byproduct, that nourishes the lining of the intestines. Meanwhile, the microbes the ones right next to the intestinal lining – it depends – they may be either promoting the lining’s health or irritating it.
ALBERTO MARTIN
Early days.







1 comment for “Alberto Martin – Carbohydrates and Colon Cancer”